Others
Categories
Featured Products
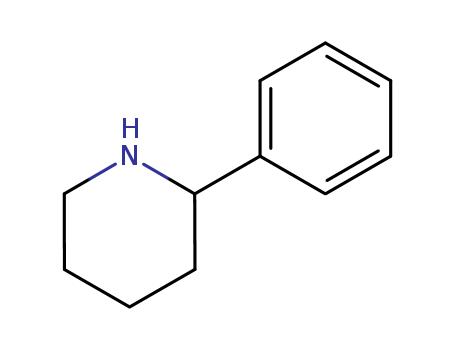
Reputable factory supply 2-PHENYLPIPERIDINE 3466-80-6 in stock with high standard
- Molecular Formula: C11H15N
- Molecular Weight: 161.247
- Appearance/Colour: clear colorless liquid
- Vapor Pressure: 0.0147mmHg at 25°C
- Melting Point: 18 °C
- Refractive Index: 1.522
- Boiling Point: 257.253 °C at 760 mmHg
- PKA: 9.74±0.10(Predicted)
- Flash Point: 110.96 °C
- PSA: 12.03000
- Density: 0.967 g/cm3
- LogP: 2.83000
2-PHENYLPIPERIDINE(Cas 3466-80-6) Usage
|
Synthesis Reference(s) |
Synthetic Communications, 25, p. 3789, 1995 DOI: 10.1080/00397919508011452Chemical and Pharmaceutical Bulletin, 43, p. 1422, 1995 DOI: 10.1248/cpb.43.1422 |
InChI:InChI=1/C11H15N/c1-2-6-10(7-3-1)11-8-4-5-9-12-11/h1-3,6-7,11-12H,4-5,8-9H2
3466-80-6 Relevant articles
Rapid Synthesis of α-Chiral Piperidines via a Highly Diastereoselective Continuous Flow Protocol
Shan, Chao,Xu, Jinping,Cao, Liming,Liang, Chaoming,Cheng, Ruihua,Yao, Xiantong,Sun, Maolin,Ye, Jinxing
, p. 3205 - 3210 (2022/05/07)
A practical continuous flow protocol has...
Zinc-Catalyzed Asymmetric Hydrosilylation of Cyclic Imines: Synthesis of Chiral 2-Aryl-Substituted Pyrrolidines as Pharmaceutical Building Blocks
W?glarz, Izabela,Michalak, Karol,Mlynarski, Jacek
supporting information, p. 1317 - 1321 (2020/12/09)
The first successful enantioselective hy...
Borane-Catalyzed Reduction of Pyridines via a Hydroboration/Hydrogenation Cascade
Yang, Zhao-Ying,Luo, Heng,Zhang, Ming,Wang, Xiao-Chen
, p. 10824 - 10829 (2021/09/08)
We have developed a method for a B(C6F5)...
Borenium-Catalyzed Reduction of Pyridines through the Combined Action of Hydrogen and Hydrosilane
Clarke, Joshua J.,Maekawa, Yuuki,Nambo, Masakazu,Crudden, Cathleen M.
supporting information, p. 6617 - 6621 (2021/09/02)
Mesoionic carbene-stabilized borenium io...
3466-80-6 Process route
-
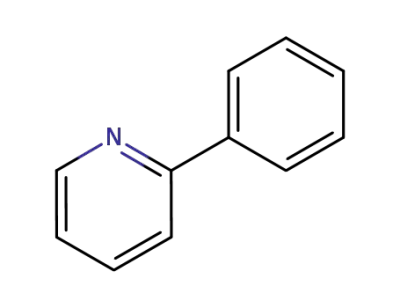
-
1008-89-5
2-phenylpyridine

-
-
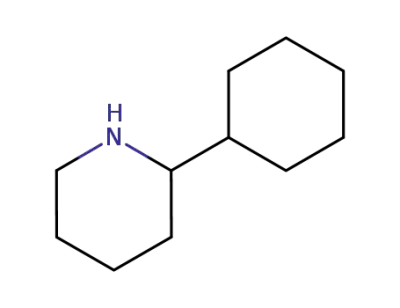
56528-77-9
(+/-)-2-cyclohexyl-piperidine

-
-
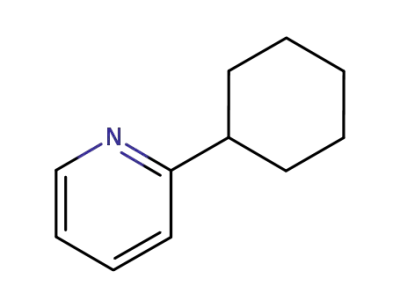
15787-49-2
2-pyridylcyclohexane

-
-
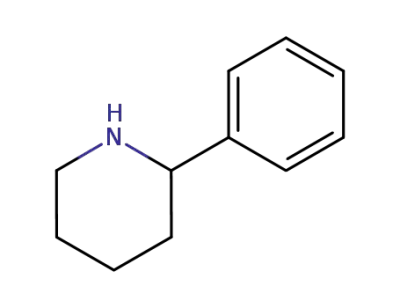
3466-80-6
2-phenylpiperidine
| Conditions | Yield |
|---|---|
|
With
Cp*Rh(2-(2-pyridyl)phenyl)H; hydrogen;
In
neat (no solvent);
at 100 ℃;
for 48h;
under 27361.8 Torr;
Catalytic behavior;
Glovebox;
|
32%
17% 39% |
|
With
hydrogen;
In
tetrahydrofuran;
at 60 ℃;
under 15001.5 Torr;
Temperature;
Catalytic behavior;
Autoclave;
Glovebox;
|
|
|
With
hydrogen;
In
isopropyl alcohol;
at 130 ℃;
for 24h;
under 22502.3 Torr;
Reagent/catalyst;
Autoclave;
|
60 %Chromat.
10 %Chromat. 22 %Chromat. |
-

-
1008-89-5
2-phenylpyridine

-

-
56528-77-9
(+/-)-2-cyclohexyl-piperidine

-

-
3466-80-6
2-phenylpiperidine
| Conditions | Yield |
|---|---|
|
With
potassium hydroxide; aluminum nickel;
In
methanol;
for 148.3h;
Title compound not separated from byproducts;
Heating;
|
83%
5% |
|
With
formic acid; [Ru(η6-C6H6)Cl(N,N-ethylenediamine)]PF6;
In
water;
at 170 ℃;
for 10h;
Autoclave;
|
3466-80-6 Upstream products
-
30091-51-1
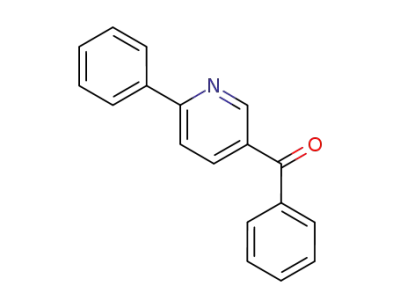
5-benzoyl-2-phenylpyridine
-
10413-00-0
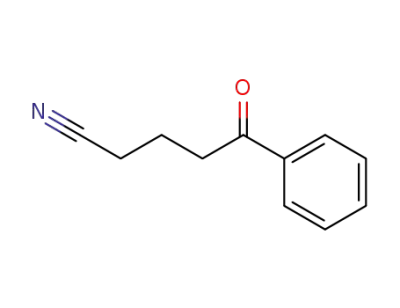
5-oxo-5-phenylpentanenitrile
-
136667-30-6
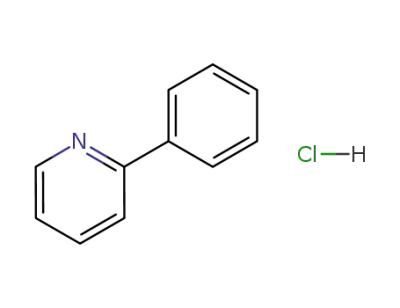
2-phenyl-pyridine; hydrochloride
-
505-18-0
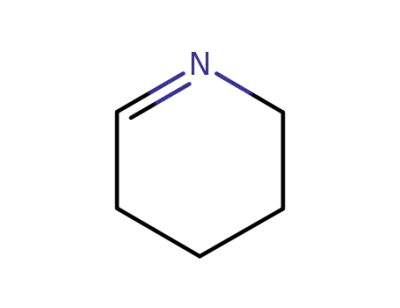
2,3,4,5-tetrahydropyridine
3466-80-6 Downstream products
-
98817-10-8
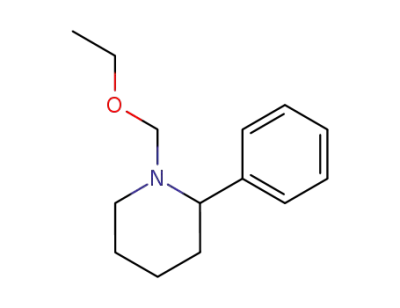
N-(etossimetil)-2-fenilpiperidina
-
139881-90-6
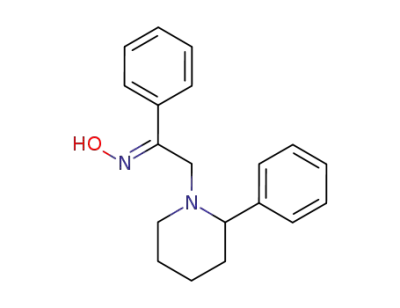
(E)-α-(2-phenylpiperidin-1-yl)acetophenone oxime
-
82902-45-2
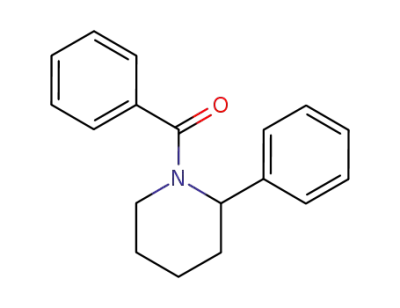
1-benzoyl-2-phenylpiperidine
-
111422-83-4
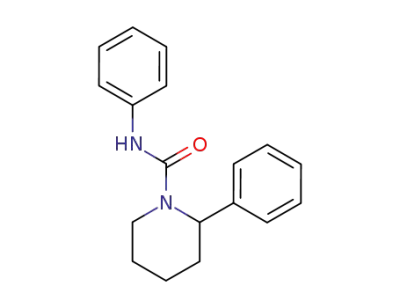
2-Phenyl-piperidine-1-carboxylic acid phenylamide